
GLP-1 receptor agonists are already reshaping people and healthcare systems for the management of obesity. Treatment paradigms in cardiovascular disease, sleep apnea and MASH are set to be disrupted, but it is not all gravy; weight loss post GLP-1s is often not maintained and not everyone can tolerate their side effects. We share our perspectives on the outlook for the space, including some of the implications for biopharma, service providers and investors.

Radioligand therapies (RLTs) are an emerging class of targeted therapies that could transform the way some cancers are treated. The RLT market is expected to grow significantly over the course of this decade, however several challenges will need to be overcome if the market is to achieve its full potential. In this white paper we consider the scientific, clinical, and commercial requirements for success.

Treatment paradigms in oncology are constantly evolving with more precise and efficacious therapies. One modality with strong potential are mRNA therapeutics which represent a significant growth area and could transform the way some cancers are treated. In this white paper we explore the potential for mRNA to transform the oncology landscape, considering the scientific, clinical and commercial requirements for success

Psychedelic-assisted therapies with MDMA and psilocybin are poised to transform the treatment options for a variety of hard-to-treat mental illnesses. However, psychedelics face regulatory and commercial headwinds, which manufacturers will need to navigate for psychedelics to achieve their full potential. Now may be a unique opportunity for investors to contribute to the development of psychedelics […]

An exploration within the growing CDMO space of manufacturing capabilities and efficiencies that support development of complex, innovative proteins / biologics, which continue to be increasingly pertinent and relevant in the market.

What are the key features of a propriety diagnostic tool that would enable isolation of a unique market opportunity? What is the business model that should be deployed to incentivize investment in this type of tool? In this paper, we will outline the answers to these questions and provide some examples of exciting technologies which may tap this potential.

Exploration of the value potential of digital therapeutics across biopharma portfolios to help companies gain a competitive edge

Current state and future trajectory of digital health including the actionable ways in which digital health can be uniquely leveraged to fill voids in the healthcare system

Key guiding principles for effective forecasting, common pitfalls, and how to use a forecast to develop better strategy

Presented at the 2019 BIO International Convention in Philadelphia, this talk by Sam Ulin, Managing Director, examines the drivers of value for innovators, with a focus on the technologies and applications that attracted significant funding in 2018

Launching a new therapy is a complex endeavor. We will review the various activities key to a successful drug launch.

How can flexibility in clinical development can be incorporated at various points along an asset’s life cycle

Navigating new challenges of market access and reimbursement for new pharmaceuticals in today's market

Recent changes in the orphan drug market access landscape and implications for pharmaceutical developers

Distinct ways that medtech companies can incorporate digital health technologies into their portfolios

Building a well thought-out plan to establish evidence of safety and of clinical utility to establish trust in a new platform

Technical and transactional assessments required to identify the right type of partnership for a biotech’s asset or platform and overall business needs
This second installment of our “Growing Up Biotech” series identifies the key factors around a pivotal question posed when launching your product: to partner or not to partner?

In the first white paper in our series “Growing Up Biotech”, we discuss the importance of communicating a compelling product story early in a biotech’s development.

PAGs play an increasingly important role in the drug development and regulatory process. Companies that truly want to be patient-centered should engage these groups early, often, and enthusiastically.
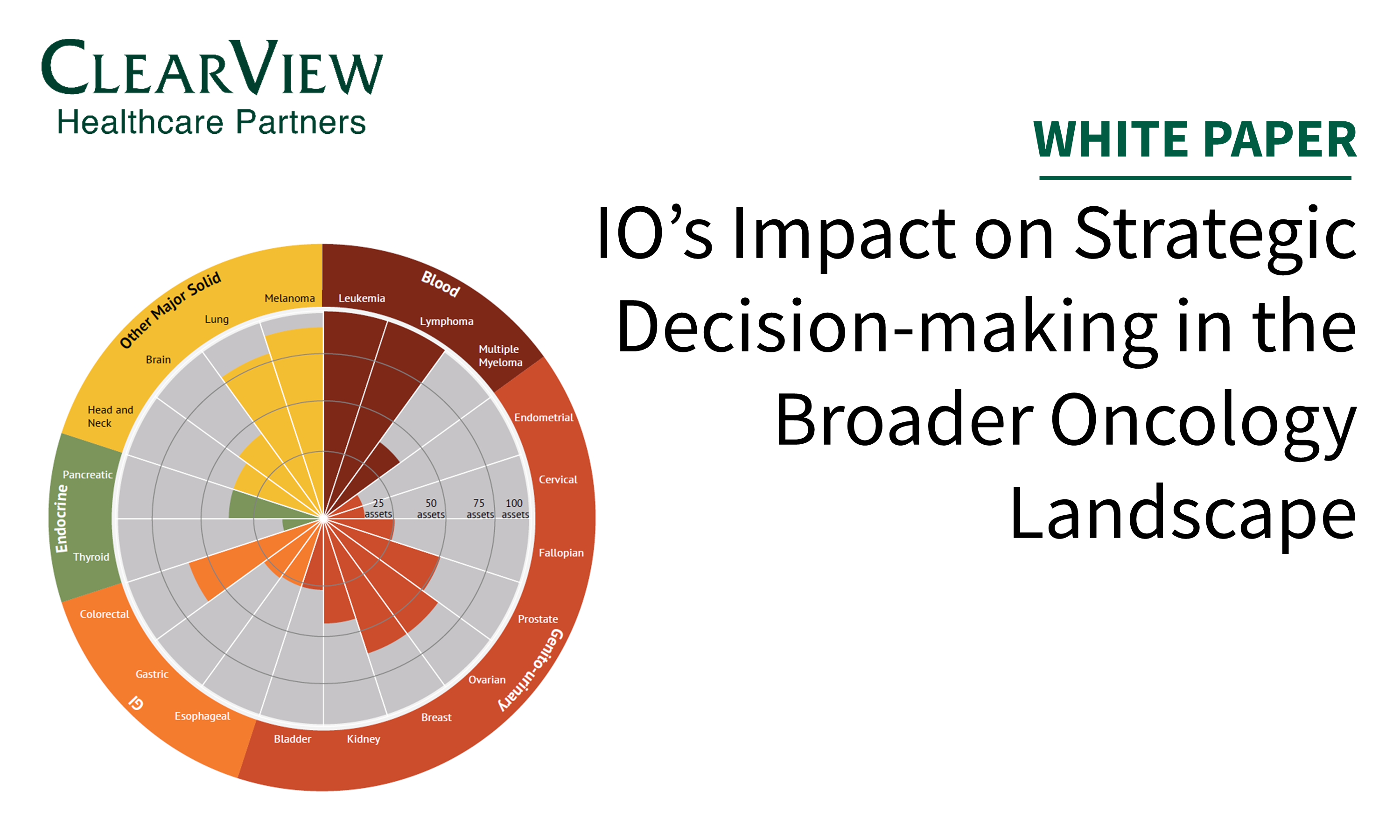
In this article published in In Vivo, we review key strategic considerations across clinical development, market access, and post-launch commercialization important for oncology drug development.

What are the clear areas of opportunity and the unique challenges associated with orphan disease drug development to help companies be best prepared to succeed in this exciting, but nuanced, space?

Informed drug development in an era of “precision medicine” requires a clear understanding of the risks and benefits that accompany the pursuit of companion diagnostics, a difficult objective given the complexity and rapid evolution of the field.

What are the key advantages and disadvantages for a biopharmaceutical company to consider prior to pursuing the breakthrough therapy pathway?